Leading AI solution for prostate cancer pathway optimization
Advancing the prostate cancer pathway with AI to support physicians, reduce costs, and deliver better care.
%
SENSITIVITY
%
SPECIFICITY
Pi™ matches the accuracy of expert radiologists
The results of a major collaborative research study with 8 UK NHS hospitals, PAIR-1, have been published in the leading journal European Radiology. This study is a landmark in the development and validation of AI for prostate cancer, showing for the first time that Pi™ operates at the level of expert radiologists working in multi-disciplinary teams, across real-world, multi-centre data encompassing different MRI scanner vendors, models and field strengths.This found that Pi™ has outstanding accuracy to detect clinically significant prostate cancer on representative, held-out validation data, with 95% sensitivity and 67% specificity. This high performance allows Pi™ to be used to help find more cancers, rule out patients who do not need further investigations, and save resources and time
What clinicians say
‘Pi can identify the most clinically significant cancers and essentially allow me to offer a same day pathway to those patients. MRi in the morning, biopsy in the afternoon
Dr Oliver Hulson
Consultant Interventional Radiologist
St James University Hospital Leeds

Trusted by our partners










Latest News
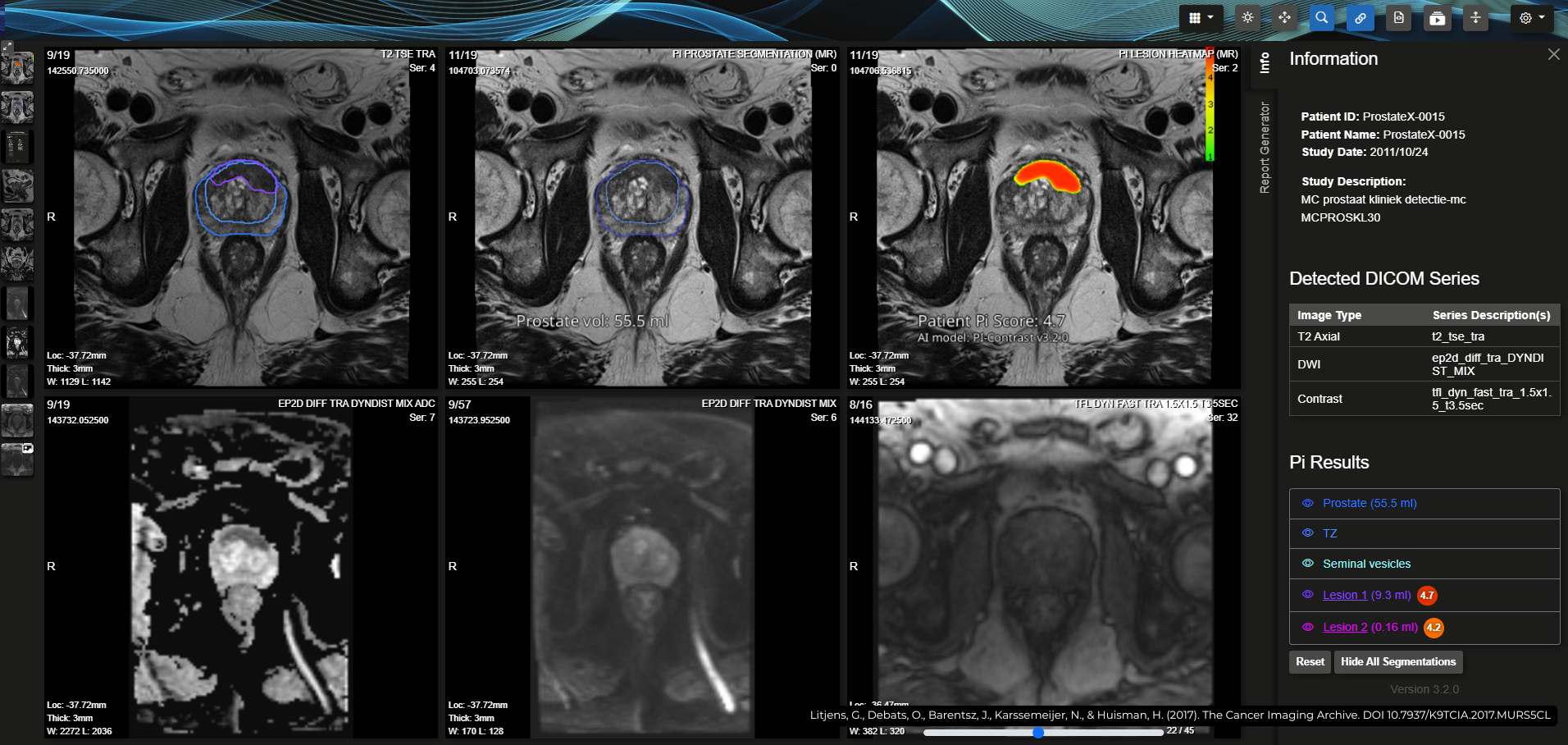
A major update released to support advanced clinical decisions

Lucida Medical raises £8.7m to power ‘same day’ cancer diagnosis

New paper published in European Radiology: AI decision support for increasing prostate biopsy efficiency

Prof. Anwar Padhani named Chief Medical Officer to guide clinical strategy

Lucida Medical, Koelis, Sectra and Hampshire Hospitals NHS Foundation Trust complete first prostate fusion biopsy using integrated diagnostic pathway

Lucida Medical has secured £2.25 million in funding from the Small Business Research Initiative (SBRI)

Lucida Medical has changed its brand identity to strengthen its position as a trusted leader in prostate cancer diagnostics
Get in touch, book a demo
To find out more, please contact us via this form